Throughout the multiple stages of storage and transport in the blood supply chain, there are many opportunities for blood to be exposed to temperatures that may render it unsafe and unusable.
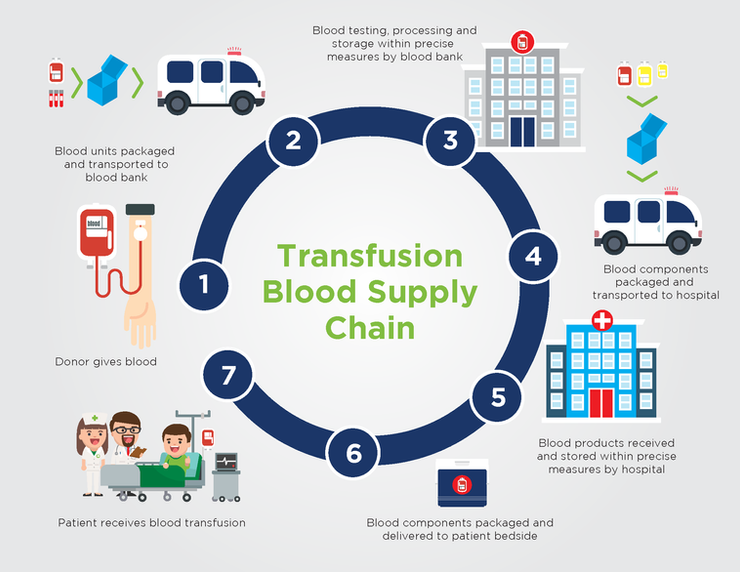
As shown in this infographic, blood and blood products must go through an elaborate series of steps before they are transfused into the patient. This is known as the transfusion blood supply chain, which can be defined as a temperature-controlled supply chain.
At each step in the blood supply chain, precise temperatures must be maintained to ensure the integrity and viability of the blood or blood products. Too cold or too warm, and the blood products may become spoiled and unusable.

Steps in the Transfusion Blood Supply Chain
- The donor gives blood.
- After donation, blood units and donor blood specimens are placed into cold storage containers and transported to the blood bank, which may be many miles away.
- At the bank, blood is tested, processed, and stored according to precise specifications determined by the blood bank in compliance with AABB standards.
- When ordered, blood products are again placed within cold storage containers and transported to the hospital or other locations where blood is needed.
- When received by the hospital, the blood products are again stored according to precise specifications determined by the hospital in compliance with AABB standards.
- When ordered by the physician, blood products are packaged and delivered to the patient’s bedside.
- Finally, the blood is transfused into the patient as needed.
Each of these 7 steps represents a possible point of failure where any delays, miscommunications, or procedural issues can cause serious problems. If any blood products must be returned, several steps on the supply chain must run in reverse following an equally strict protocol [1]. Again, any failures in the protocol can mean wasted products.
Wasted Blood is Costly
Blood products represent an expensive and labor-intensive resource, accounting for approximately 1% of hospital expenditures [2]. Yet the transportation and storage of blood products is often an inefficient and costly process. For example, red blood cell (RBC) product wastage in hospitals is reported to range anywhere from 0.1% to 6.7%.
In one study, the annual direct cost of intraoperative RBC wastage at Vanderbilt University Medical Center (VUMC) amounted to approximately $249,314 in 2010, using an estimated direct cost of $225.42 per unit of leukoreduced RBCs. This figure does not account for the overhead costs associated with the procurement, management, storage, and issue of these products [3].
“According to the Journal of Clinical Oncology, the cost to have a blood transfusion can range from $1,800 to $3,000 per unit of RBCs transfused. The acquisition cost of a unit of RBCs is approximately $200” [4]. This cost does not include the indirect costs of labor and reagents when preparing a unit of RBCs for transfusion, which is lost since the unit cannot be reissued to another patient.
Not only are blood products costly, but unnecessary losses or spoilage can result in shortages and put human lives at risk. A more reliable and cost-effective solution is needed.
Learn More:
Find out the three main drawbacks of how blood is handled today here.
Sources
1: Standards for Blood Banks and Transfusion Services, 30th edition. AABB. April 1, 2016. For current edition of AABB Standards (31st edition).
2: Supply chain management of blood banks. Pierskalla, WP. In: Brandeau, ML.; Sainfort, F.; Pierskalla, WP., editors. Operations research and health care: a handbook of methods and applications. Boston (MA): Kluwer Academic; p. 103-45.
3: National Blood Collection & Utilization Survey [Internet]. Washington (DC): US Department of Health & Human Services; 2011.
4: Implementation of a new blood cooler insert and tracking technology with educational initiatives and its effect on reducing red blood cell wastage. Transfusion. Fadeyi, Emmanuel A., et al. 2017;57(10):2477-2482.
MaxPlus Thermal Control Transport Systems